
For the acute panel, if either IgG or IgM screen is positive, there is automatic reflex to Western blot for IgG and IgM. Some commercial laboratories provide reflex profile testing for Lyme disease. Draw volume changed from 3.0 mL to 1.5 mL. These include syphilis, subacute bacterial endocarditis (SBE). 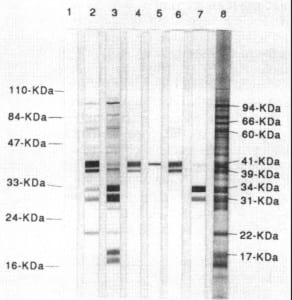
burgdorferi IgG+IgM with reflex to immune blot panel (S) Consumer Name Alpha Borrelia burgdorferi Ab.IgG+IgM with reflex to immune blot panel, Blood Basic Attributes Class PANEL.MICRO Type Laboratory First Released Version 2. Mayo previously used IFA for the IgM portion effective. If the Western Blot is negative, causes of false positive immunoassays should be considered. Method Additional Names Short Name B burgdor IgG+IgM W reflex to IB pnl Ser Display Name B. Updates: : Mayo now uses Western Blot methodology for both IgG and IgM confirmations. *Reflex for positive or equivocal Lyme tests are referred to reference laboratory for Western blot analysis. retested by separate IgM and IgG immunoblots (Western blot). Test Name: LYME DISEASE SEROLOGY EVALUATION REFLEX, SERUM General Information Lab Order Codes: LYMS, LYCON. Other details on the tests are as follows: Test name All three tests use red top (serum) tubes. Testing will be run Monday through Friday on the evening shift. 
Testing detects infection caused by both European and North American strains of Borrelia. A Lyme IgM test should be used for patients with signs and symptoms that are consistent with Lyme disease. Lyme disease is caused by a bacterium, Borrelia burgdorferi, that is transmitted by the ticks of the genus Ixodes. No testing is recommended for patients with erythema migrans. Qualitative determination of IgM/IgG antibodies by screen with reflex to Lyme Western Blot testing if screen is positive. The CDC does not recommend performing Lyme IgM Western Blot testing on patients with signs or symptoms of greater than 30 days’ duration, rationalizing a different approach to confirmatory testing for acute and late disease. Lyme Disease and Helicobacter IgG Testing now available in Core LaboratoryĮffective today, the University of Iowa Hospitals and Clinics core clinical chemistry laboratory will being running assays for Lyme Disease antibodies and Helicobacter pylori IgG antibodies.įor the Lyme Disease testing, we will continue offering options for acute disease (less than four weeks from onset of disease symptoms) or chronic disease as was offered previously with mailout testing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
